LATEST
NEWS:
January 2024 : Our last review paper have been accepted in Adv. Synth. Catal. "Visible light driven cyclizations" Congratulations to all participants!!!!
January 2024 : Happy NEw Year!!!!
October 2023: Our first postdoc in Khalifa University start working. Welcome Dr. Shaista Saitan, We wish you all success!!!
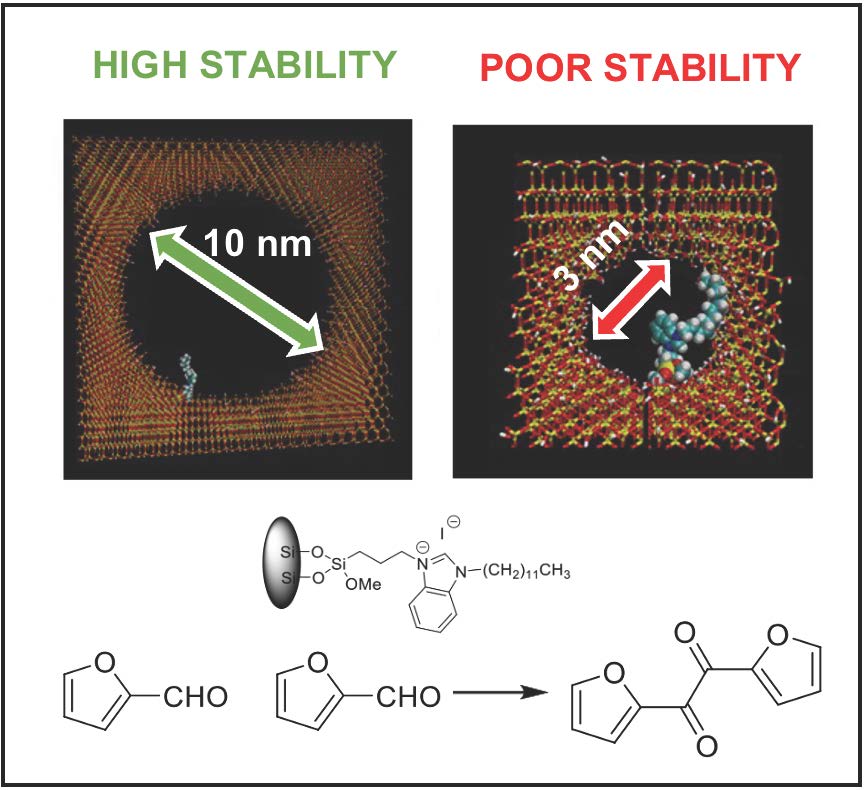
October 2022: A New paper from our European Project Multhycat!!!! We published our efforts in benzoin condensation in Chemistry European Journal. Congratulations to everybody involved!!!!
August 2022: We moved to Khalifa University taking a full Professor position!!!! We hope a great stay there!!!!
March 2022: Our Spiro book is finnally online!!! Thanks to all co-authors. GRAT JOB|||||

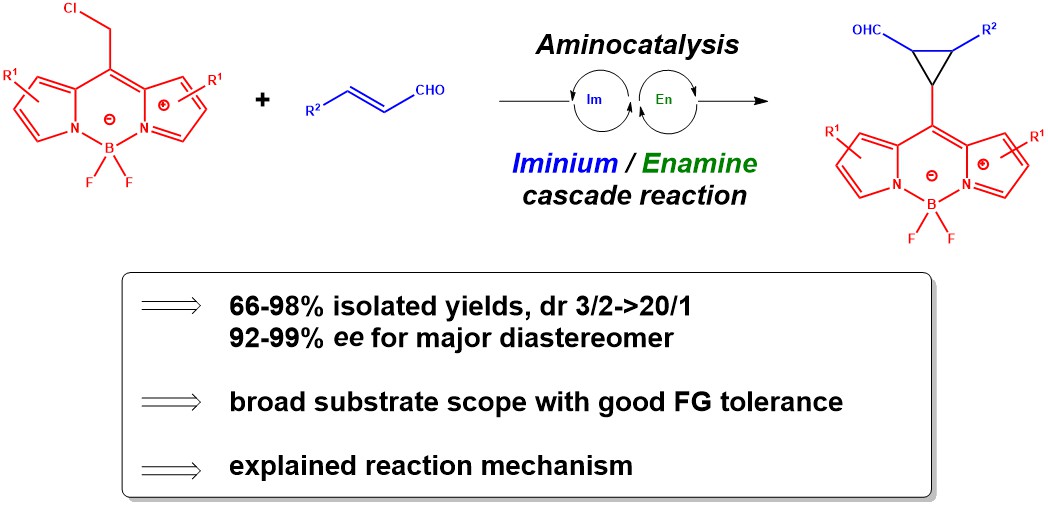
December 2021: Our last paper in Chiral BODIPYS is online: Stereoselective Cyclopropanation of Boron
Dipyrromethene (BODIPY) Derivatives by an
Organocascade Reaction. Congratulations to Jan and Jan's group!!!!!
October 2021: One of our papers have been selected as one of the best organocatalytic papers published in Chemical Science to celebrate the Nobel Prize in Organocatalysis:
Proline bulky substituents consecutively act as steric hindrances and directing groups in a Michael/Conia-ene cascade reaction under synergistic catalysis.Chem. Sci. 2019, 10, 4107-4115.
October 2021: It is a great day for organocatalysis. Professors Benjamin List and David MacMillan win the Nobel Prize!!!! Congratulations a well deserved Prize!!!!
April 2021: Our work with BODIPYs have been highlighted in Organic Process Research and Development (ACS journal). Thanks for the interest, we hope to develop new reactions soon!!!!
March 2021: Our last collaboration with Professor Jan Vesely's group and Professor Patil have been published!!! Congratulations to all!!!!
February 2021: Our latest work is published in Chemical Science. Synthesis of Chiral Bodipys through Organocatalysis. Great collaboration with Professors Luis Crovetto and J.M. Cuerva research groups (University of Granada), they are the wizards behind fluorescence and photoluminiscence. Congratulations Marta and the rest of the team
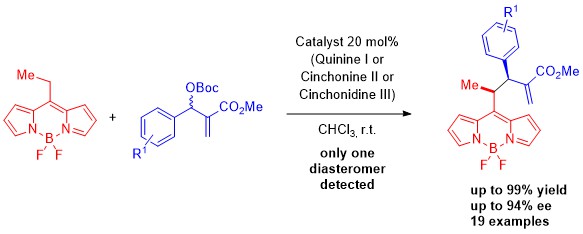
January 2021: Our first COVID paper!!!! and the first paper with Marta as Corresponding Author it has been published in Advanced Synthesis and Catalysis: CONGRATULATIONS!!!
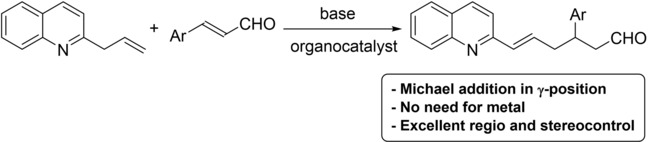
September 2020: Our Collaboration with Raja's group render another paper. Congratulations Cameron!!!:
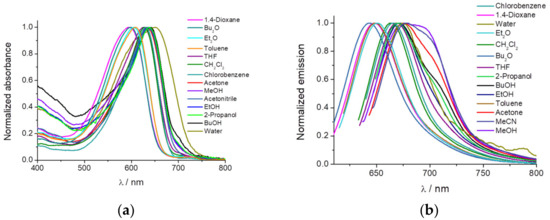
November 2019: Great collaboration with University of Granada (Professor Luis Crovetto) for the study of fluorescence properties of new molecules. It have been published in Sensors. Great job!!!
November 2019: Our collaborations are going well. 2 more papers from our collaboration with ITQ, University of Piamonte Orientale and Professor Robert Raja's research group (UoS) regarding the synthesis and applications of Multyhibrid catalysts.
July 2019: Marta Meazza win the Dean's Award for her research contributions. Congratulations Marta!!!! A well deserved award!!!
April 2019: Our latest paper in Synergistic Catalysis is online.
Congratulations to Marta and Sara!!!!
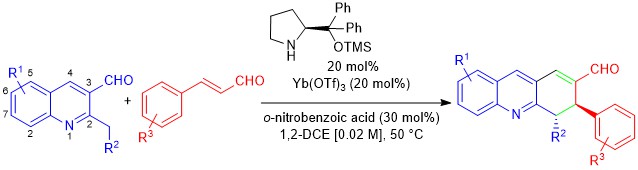
March 2019: Our latest paper in Synergistic catalysis is online.
Proline bulky substituents consecutively act as steric hindrances and directing groups in a Michael/Conia-ene cascade reaction under synergistic catalysis.
Congratulations to Marta, Jan and Raquel's group!!!!
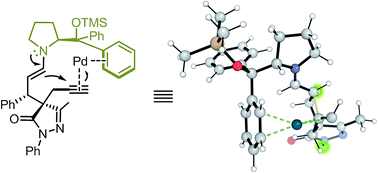
July 2018: Our latest review is online. Synthesis of Lactams by tandem reactions. Congratulations to Marta and Xavi!!!!
July 2018: Our latest work in Synergistic Catalysis is online. Activation of acetyl pyridines to synthesise highly substituted cyclohexenes. Congratulations Marta, Gabi, Kai and Andrea's group.
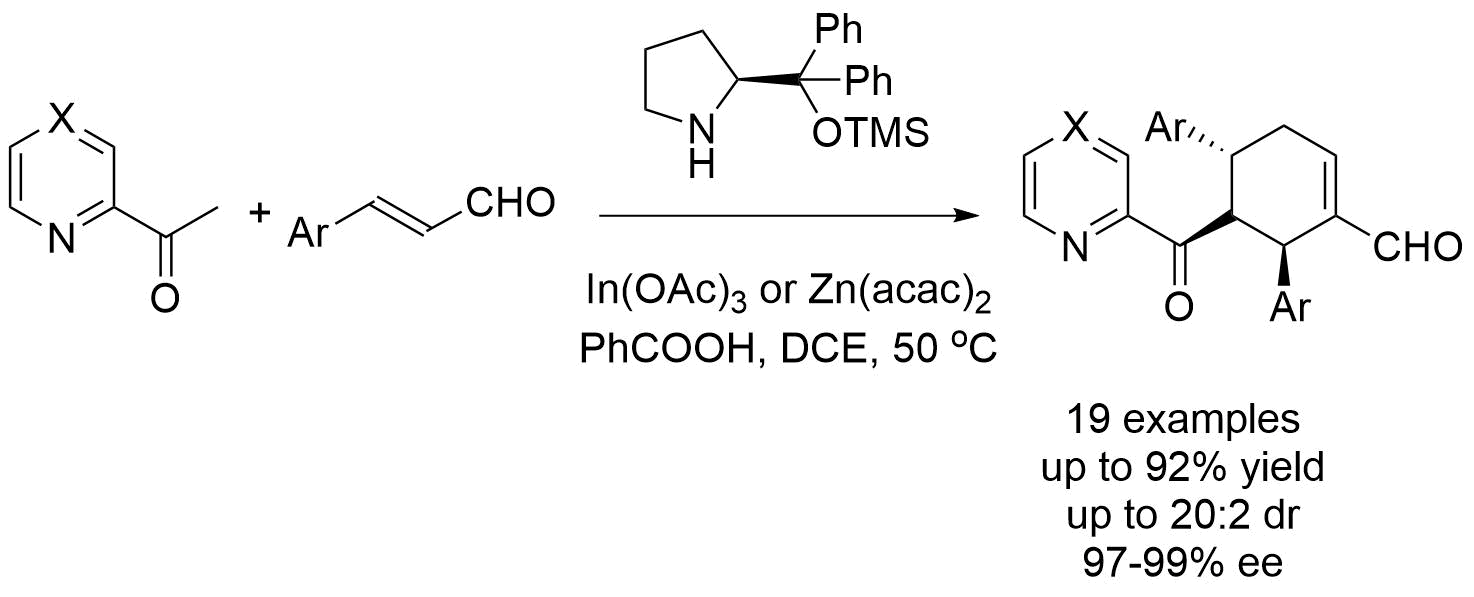
July 2018: Our latest work in Synergistic Catalysis is online. The synthesis of spiropyrazolones through a formal ring contraction is published in Chemical Science. Congratulations Marta, Jan´s and Jana´s group!!!!
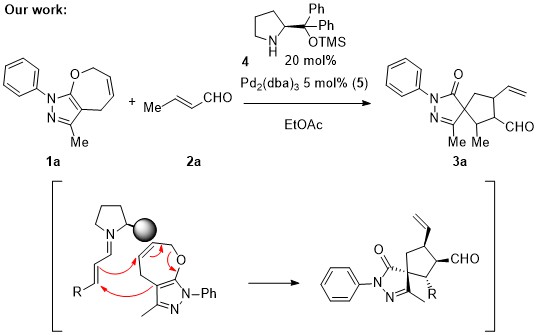
July 2018: Our latest Review is online. The synthesis of spirocompounds is published in Chemical Society Reviews. Congratulations Marta, Aishun, Yang and Hao!!!!
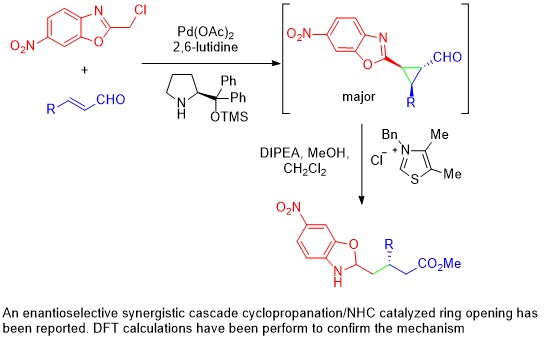
November 2017: Our latest work in Synergistic Catalysis is online. Congratulations Marta. Victor and Pedro.
November 2017: Our work regarding the synthesis of spirolactams have been highlighted in Organic Chemistry highlights!!!!
May 2017: A funded PhD position in our group is open for EU/UK students. The PhD will deal with the synthesis of new polymers for optoelectronic applications and asymmetric synthesis.
To apply: https://jobs.soton.ac.uk/Vacancy.aspx?ref=853517EB
February 2017: Our review regarding the uses of vinyl cyclopropanes is online, Congratulations Marta.
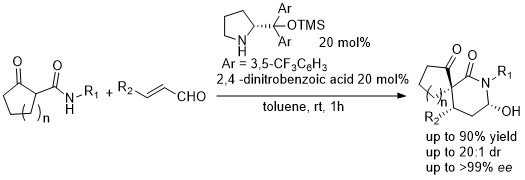
February 2017: Our last paper in organocatalysis is online, Congratulations Kai, Marta and Jan's group.
January 2017: Our last paper in organocatalysis is online, Congratulations Marta and Mike.
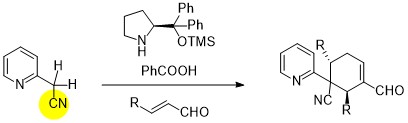
November 2016: Our last paper in synergistic catalysis is online, Congratulations Kai, Marta, Clotilde and Anabel.
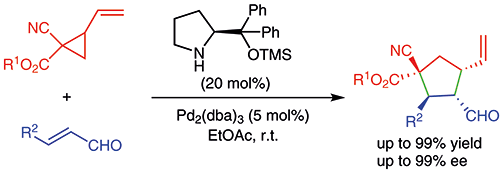
May 2016: Marta Meazza got an Honorable Mention in the ABTA Doctoral Researcher Awards. Congratulations, A well deserved prize for your exceptional work!!.
May 2016: Our last paper in Synergistic Catalysis is online. Congratulations Marta!!
May 2016: After finishing her PhD Dr. Marta Meazza started a postdoc in the group of PRofessor Karl Anker Jorgensen in Aarhus. Congratulations and good luck!!!
April 2016: Our last paper in organocatalysis a Feature Article in The Journal of Organic Chemistry is online. Synthesis of cyclopropanes using benzyl chlorides. Congratulations Marta, Maria and Yang's group!!

February 2016: Marta Meazza get her PhD degree!!!! Well done!!! Great Job!!!
February 2016: Our last paper in photocatalysis is online. Synthesis of phophoramidates using organic dyes. Congratulations Marta, Agga and Luke!!
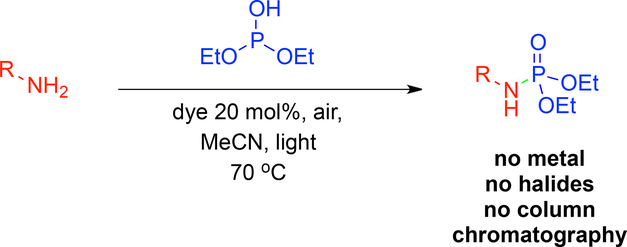
January 2016: Our Review in Synthesis is online: Merging transition-Metal activation and Aminocatalysis. Congratulations Marta!!
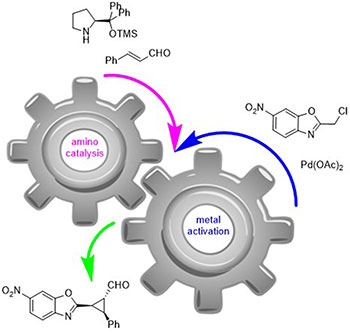
January 2016: Our last paper of Photocatalysis has been accepted in ChemistrySelect. Great job Luke, Victor and Marta!!!!
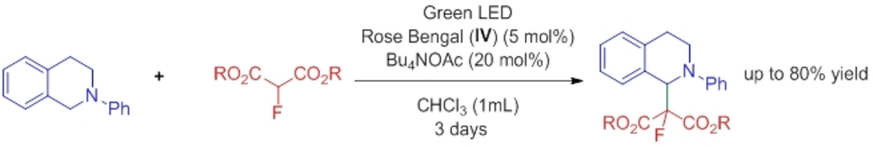
October 2015: Our last paper of Synergistic catalysis is accepted in Chemical Science. Great job Marta!!!!
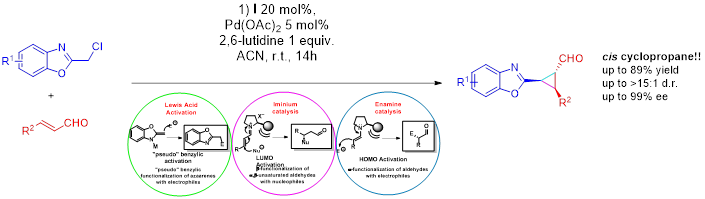
October 2015: A new paper have been published. Our collaboration with Professor Hao Guo (Fudan University) is working very well and here is our second common paper in Tetrahedron Letters. Congratulations to Guo's group!!!!

October 2015: Marta Meazza have been selected for a poster presentation at the Royal Society of Chemistry's Organic Division Poster Symposium 2015!!!! Congratulations!!!
October 2015: Our last paper about anthrones has been accepted in Scientific Reports. Congratulations Victor, Marta, Greg, Jiri and Jan!!!!!
May 2015: Lecture in East China Normal University. Visiting my good friend Professor Jian Zhao!!! Great time in China and remembering our postdoc with Professor Benjamin List!!!

May 2015: Our latest Molecules is online. Congratulations Victor, Tope and Marta!!!! May 2015: Our latest Molecules is online. Congratulations Victor, Tope and Marta!!!!
“Highly Diastereoselective Synthesis of Spiropyrazolones”
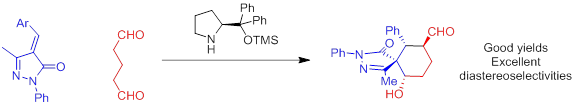
April 2015: Marta Meazza have been selected to give a talk at the 26th SCI Regional Postgraduate Symposium at Southampton on Novel Organic Chemistry. http://www.soci.org/Events/Display-Event?EventCode=FCHEM451 Congratulations Marta!!!
March 2015: Our article "Synergistic Catalysis: Enantioselective Addition of Alkylbenzoxazoles to Enals " (Chem. Eur. J. 2014, 20, 16853-16857) have been highlighted in SYNFACTS. Congratulations Marta and Victor!!!!
January 2015: Our article "Enantioselective Organocatalytic Synthesis of Fluorinated Molecules" (Chem. Eur. J. 2011, 17, 2018–2037) is among the top 20 most cited Reviews in Chemistry—A European Journal of the past 20 years!!! Congratulations Guillem and Xavi!!!!!
October 2014: Our latest Chemistry European Journal is online. Congratulations Marta and Victor!!!
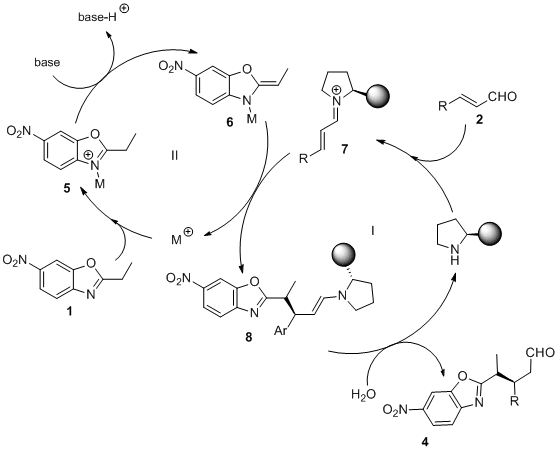
In good company: A novel catalytic enantioselective methodology based on synergistic catalysis is reported. The strategy involves: 1) the metal-Lewis-acid activation of alkylazaarenes, and 2) the secondary-amine activation of enals (see scheme). Consequently, highly functionalized chiral alkylazaarenes were obtained in good yields and with reasonable stereoselectivity.
October 2014: Nice Review about our organocatalysis book in Angewandte
September 2014: Our latest paper on Synergistic Catalysis have been accepted in Chemistry European Journal!!! Congratulations Marta and Victor.
September 2014: A new paper have been published. Our collaboration with Professor Robert Raja is working very well and here s our first common paper in Catalysis Science and Technology: Congratulations to Raja's group!!!!
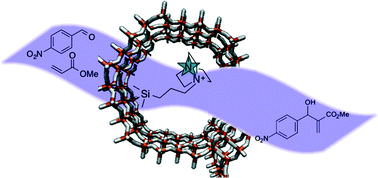
August 2014: Marta and Victor have attended to the ACS Meeting 2014 in San Francisco where they presented their lasts works.
May 2014: Our latest Chemical Communnications is online. Congratulations to Victor, Marta and Piotr!!!
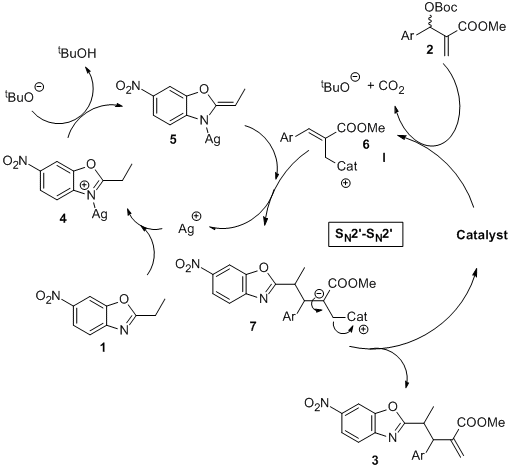
An expedited method has been developed for the diastereoselective synthesis of highly functionalized alkyl-azaarene systems with good yields and high diastereoselectivities (> 15:1 dr). The methodology includes a synergistic catalysis event involving organometallic (10 mol% AgOAc) activation of an alkyl azaarene and Lewis base (10 mol% DABCO) activation of a Morita–Baylis–Hillman carbonate. The structure and relative configuration of a representative product were confirmed by X-ray analysis
March 2014: Satoshi Okusu from Shibata's group has started a one month placement in our research group. Welcome!!!
February 2014: Dong Wang and Hun Yi Shin from Yang's group have started a one month placement in our research group. Welcome!!!!
January 2014: Our latest Advance Synthesis and Catalyst is online. Congratulations to Yang's team and Bing
Abstract

Highly enantioselective cascade reactions for the synthesis of fluoroindanes and chromanols derivatives are described. The cascade reactions consisted of either a double Michael reaction or Michael–hemiacetal formation via the addition of fluorobis(phenylsulfonyl)methane (FBSM) to enals. The final products were obtained in good yields with excellent stereoselectivities.
November 2013: Xavi received a Marie Curie Intraeuropean fellowship for a postdoc in Imperial College (London).
Congratulations!!!! All our PhD students to date have received a Marie Curie fellowship after working with us!!! Congratulations Xavi Companyo and Andrea-Nekane R. Alba!!!!
November 2013: A New Review from our group is online:
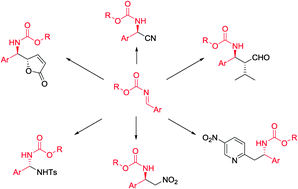
Nucleophilic addition to carbon–nitrogen double bonds (imines) represents one of the most common strategies for the synthesis of amine derivatives. In order to circumvent the problem associated with low reactivity of imines in nucleophilic addition, various imines with electron-withdrawing groups at nitrogen have been studied, and many of them were successfully applied in asymmetric methodologies. Especially N-carbamoyl imines were found to be useful in the enantioselective synthesis of various organic compounds, due to their increased reactivity toward nucleophiles as well as limited difficulties connected with the removal of the carbamoyl moiety in target molecules. The aim of this review is to cover enantioselective methods based on N-carbamoyl imines, focusing on synthetically useful protocols.
November 2013: Our latest Tetrahedron paper is online:
An organocatalytic enantioselective synthesis of α-methylene-γ-lactams has been developed. The reaction between protected 2-aminomalonates and Morita–Baylis–Hillman carbonates is catalyzed by chiral Lewis bases to afford the corresponding lactams in excellent yields and moderate to good enantioselectivities, after work-up.
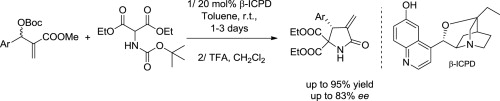
Congratulations: Xavi and Pierre!!!!!
November 2013: Our paper in the Asian Journal of Organic Chemistry: Enantioselective Organocatalytic Amination of Pyrazolones. Has been one of the top 25 most dowloaded papers!!!
November 2013: Our latest Tetrahedron Letter paper is online:
A new three-component cascade reaction for the synthesis of thiohydantoins is reported. The reaction between α-amino esters, nitrostyrenes, and aromatic isothiocyanates is efficiently promoted by organic bases to afford highly substituted thiohydantoins in moderate to good yields and diastereoselectivities.
Congratulations Victor, Kane and Marta!!!!
August 2013: Our book is published:

January 2013: Our latest Chemical Communications paper is online:
All in one pot: an organocatalytic highly enantioselective synthesis of α-methylene-γ-lactones has been reported. The reaction between protected 2-hydroxymalonates and MBH carbonates is simply catalysed by chiral Lewis bases affording after acid treatment the corresponding lactones in excellent yields and enantioselectivities.
Congratulations Xavi!!!
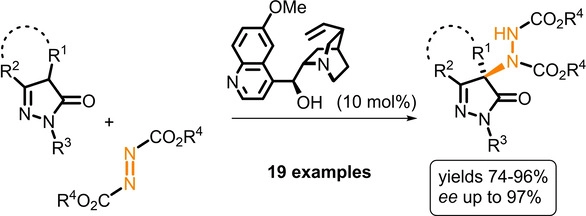
December 2012: Our latest Asian Journal of Organic Chemistry is online:

Easy as py: The organocatalytic enantioselective amination of pyrazolones is reported. The reaction between pyrazolones and diazodicarboxylates is simple and is catalyzed by quinine to afford the final aminopyarazolone derivatives in excellent yields and good enantioselectivity.
Congratulations: Jan and Marek!!!!
August 2012: We have moved at University of Southampton in August 2012.